Indications and functional outcome of the use of integra® dermal regeneration template for the management of traumatic soft tissue defects on dorsal hand, fingers and thumb | Archives of Orthopaedic and Trauma

Indications and functional outcome of the use of integra® dermal regeneration template for the management of traumatic soft tissue defects on dorsal hand, fingers and thumb | Archives of Orthopaedic and Trauma

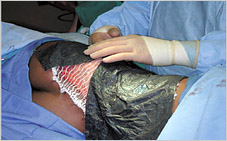
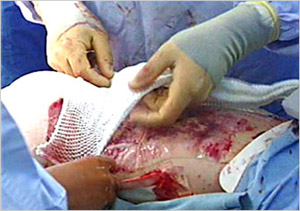
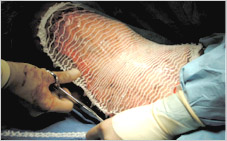
The use of the artificial dermis (Integra) in combination with vacuum assisted closure for reconstruction of an extensive burn scar--a case report. | Semantic Scholar

Figure 2 from The use of the artificial dermis (Integra) in combination with vacuum assisted closure for reconstruction of an extensive burn scar--a case report. | Semantic Scholar
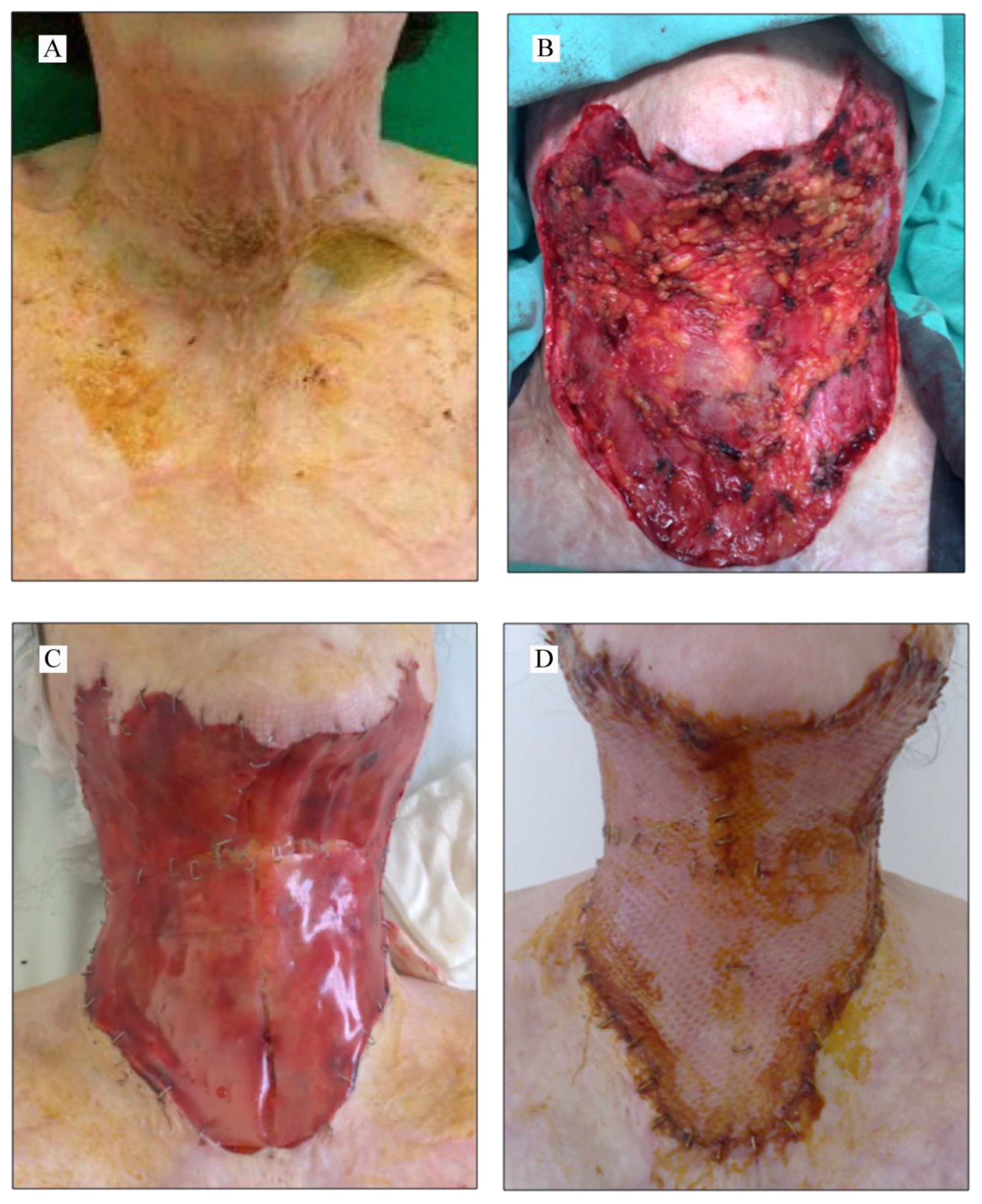
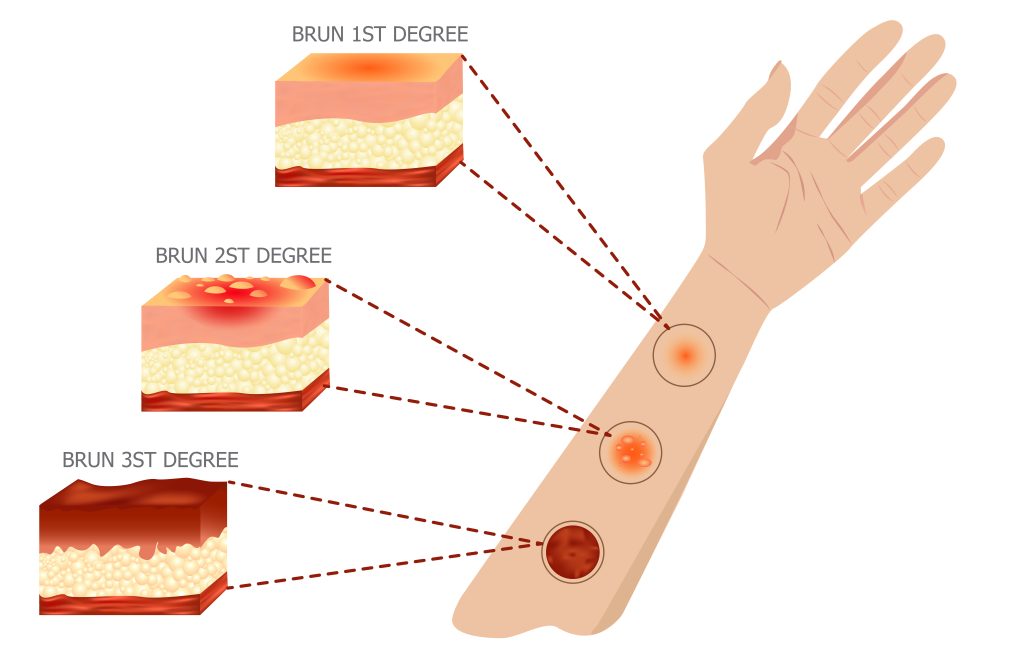
Applied Sciences | Free Full-Text | The Use of Collagen-Glycosaminoglycan Biodegradable Matrix (Integra®) in the Management of Neck Postburn Hypertrophic Scars and Contractures

Integra Lifescience Medihoney Paste Dressing, 0.5 fl oz Tube, 10/bx 31505 : Industrial & Scientific - Amazon.com

Combination of negative pressure wound therapy (NPWT) and integra dermal regeneration template (IDRT) in the lower extremity wound; Our experience with 4 cases - ScienceDirect
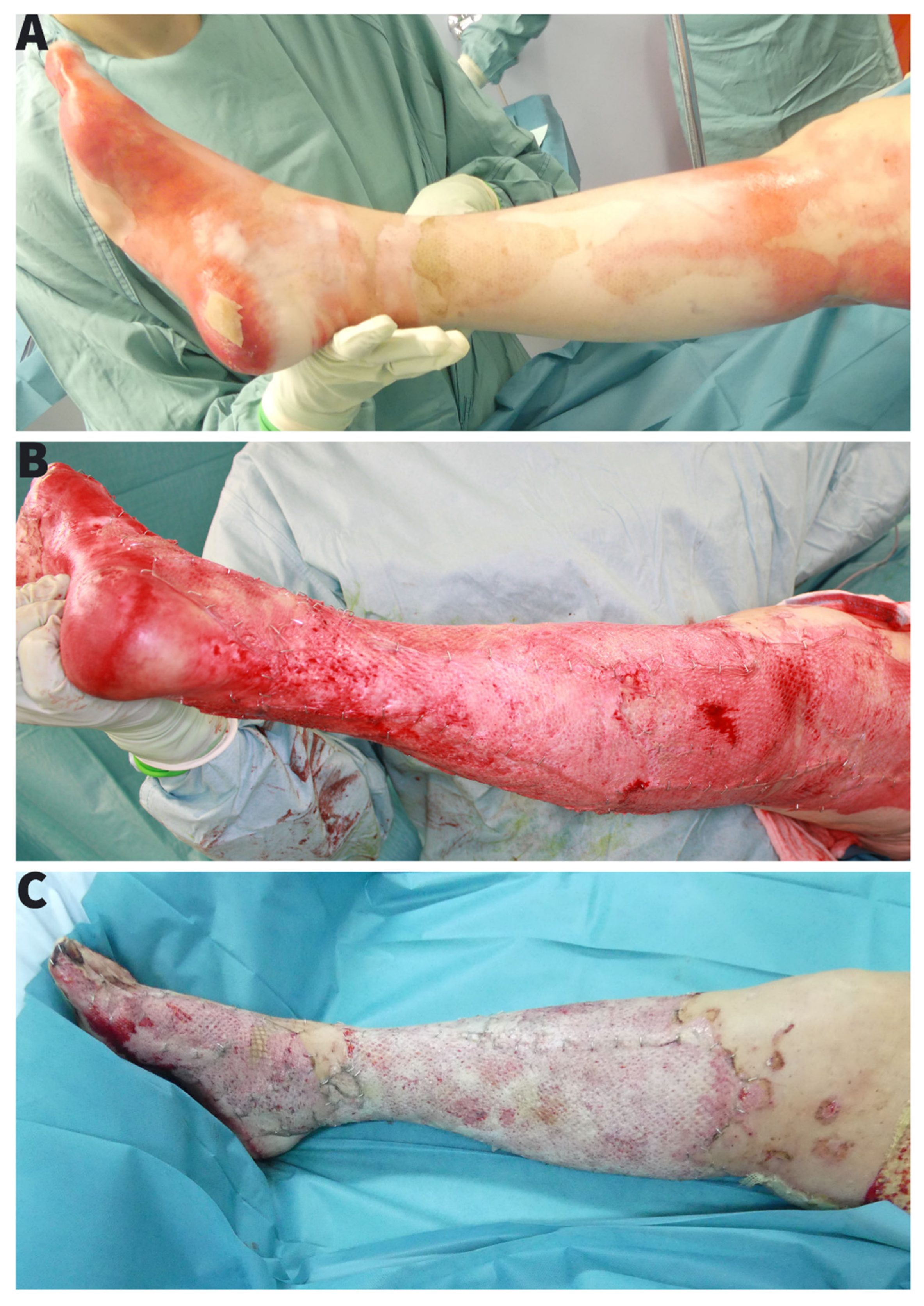
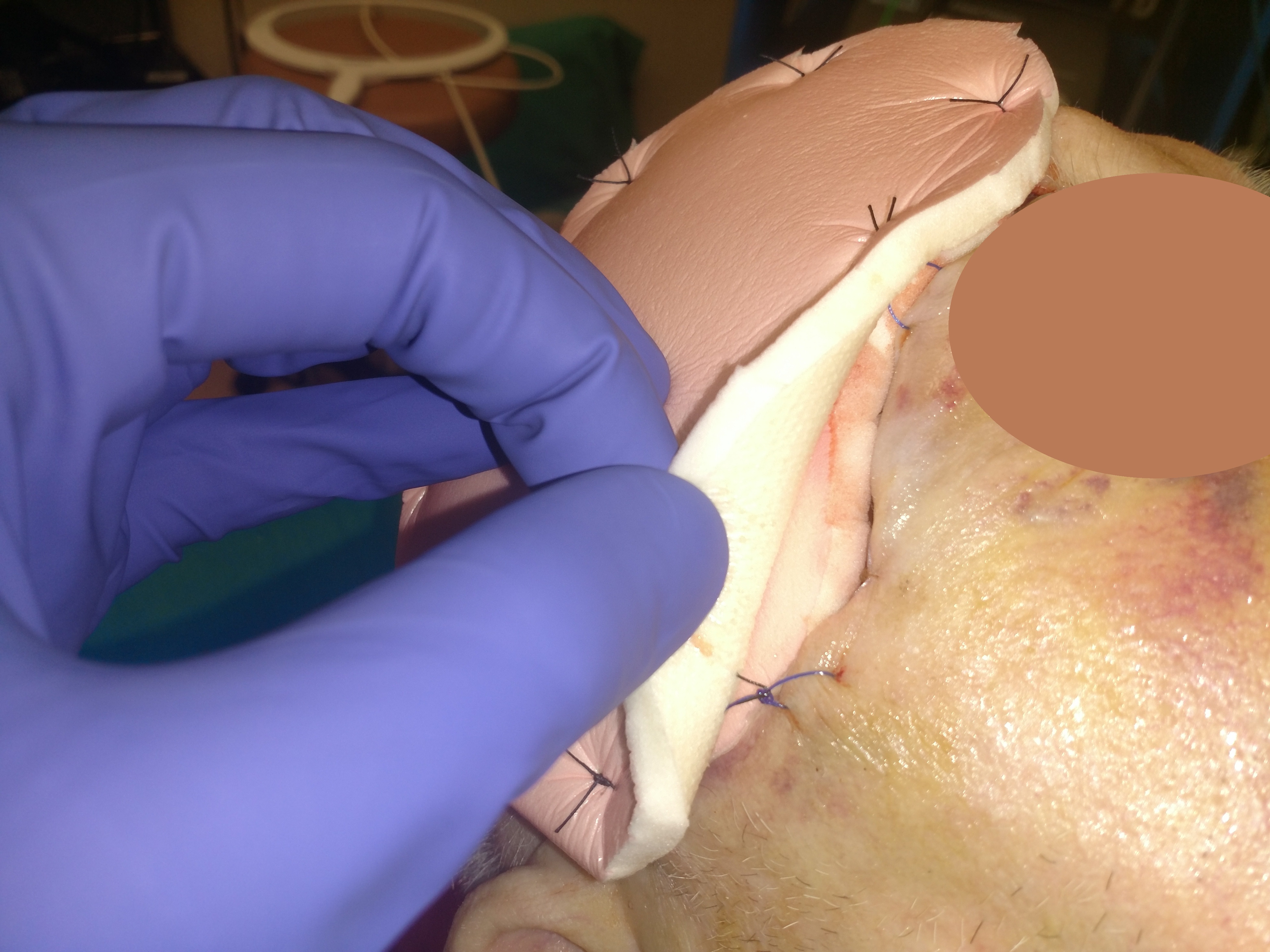
EBJ | Free Full-Text | Limb Salvage through Intermediary Wound Coverage with Acellular Dermal Matrix Template after Persistent Pseudomonas Aeruginosa Infection in a Burn Patient

Integra Lifesciences Medihoney Gel Dressing, 1.5 fl oz Tube, 31815 12EA/CA : Amazon.sg: Industrial & Scientific

RBCP - Use of dermal matrix and negative pressure dressings for the treatment of contractures in burn patients

Integra Lifescience Medihoney Paste Dressing, 1.5 fl oz Tube, Applicator 31515 | To Your Door Medical
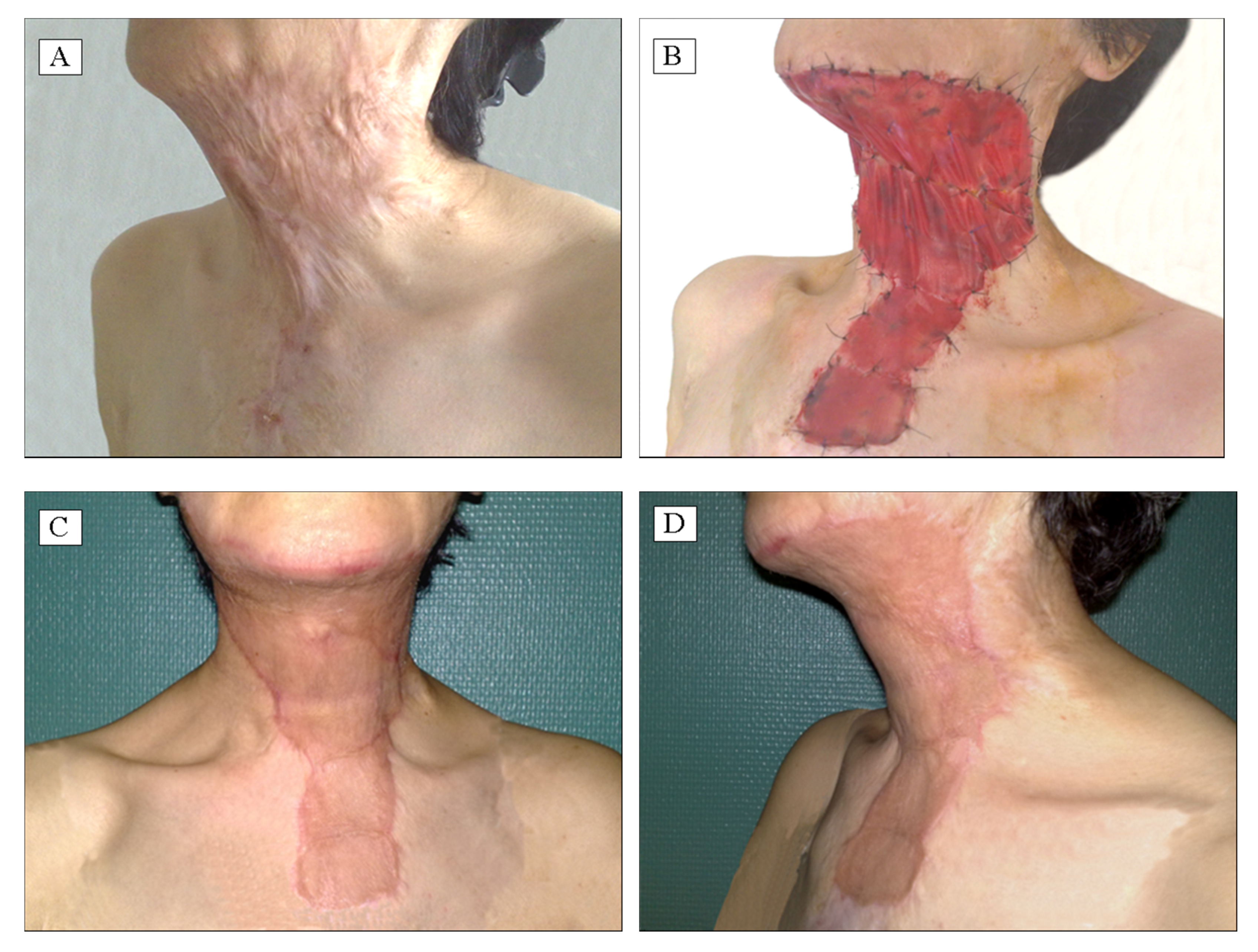
Applied Sciences | Free Full-Text | The Use of Collagen-Glycosaminoglycan Biodegradable Matrix (Integra®) in the Management of Neck Postburn Hypertrophic Scars and Contractures

Photos of a patient undergoing integra-based reconstruction of a large... | Download Scientific Diagram

Novel use of a flowable collagen–glycosaminoglycan matrix (Integra™ Flowable Wound Matrix) combined with percutaneous cannula scar tissue release in treatment of post-burn malfunction of the hand—A preliminary 6 month follow-up - ScienceDirect